- Fernando Huyke-Hernández, Research Fellow – TRIA Orthopedic Institute
- Megan Sorich, Assistant Professor Orthopedic Surgery – UT Southwestern Medical Center
- Julie Switzer, Associate Professor – University of Minnesota
Abstract
Objective
Malnutrition is associated with worse hip fracture outcomes. Optimal nutritional interventions for these patients have yet to be identified. Regarding nasogastric tube (NGT) feeding, prior studies have shown promise yet have been limited and difficult to implement. We conducted a pilot study to assess feasibility and describe outcomes.
Methods
This was a prospective longitudinal pilot study conducted in a single level 1 trauma center from June 2019 to January 2020. Patients aged 65 or older that suffered acute hip fracture due to low-energy trauma and required surgical intervention within 48 hours were included. Patients were divided into two arms: patients receiving standard of care with regular calorie counts versus patients receiving an NGT intraoperatively and subsequent early tube feeding. Feasibility was assessed via interviews/comments and recorded incidents of patient unwillingness to participate in intervention as well as interventional discontinuation.
Results
Only 16 patients (7 intervention, 9 control) were recruited after the study was ceased due to futility. The majority of patients declined the intervention due to stigma regarding NGT placement and tube feeding. All patients had their NGTs removed within 11 days after surgery. Most removals were due to tube intolerance and patient requests.
Conclusion
Implementing a prospective trial comparing hip fracture patients receiving NGT feeding and those receiving standard of care can be challenging. Identified barriers include patient unwillingness to participate due to stigma, high frequency of tube discontinuation, difficulty attaining sample , and lack of standardization or training. Future study attempts should account for barriers such as stigma, tube removals, and study and/or environment.
Keywords
hip fracture; tube feeding; nasogastric tube; early enteral feeding; stigma; barriers; prospective study
Introduction
It has been estimated that 6.3 million hip fractures will occur annually worldwide by 2050, with currently more than 300,000 hip fractures occurring annually in the United States[1-3]. In the United States, each hip fracture admission and readmission respectively cost more than $16,000 and $14,000, leading to an overall estimated $12-15 billion annually[1,4-6]. Despite advancements in surgical and medical optimization, one-year mortality is still reported at up to 30%[7]. Short- and long-term functional deterioration is also commonplace for these patients. The impact of this devastating injury at the patient and societal level has led to increasing efforts in mitigating adverse outcomes.
Malnutrition is a factor that has been associated with worse outcomes after hip fracture. A hip fracture patient’s body is in a catabolic, inflammatory state that requires compensatory nutritional intake[8]. Because older adult patients have been found to intake less than required compared to their physiologic need, they are at high risk of loss of weight, muscle mass, and fat stores, which diminishes their ability to recover and impacts outcomes[8,9]. The mortality in malnourished hip fracture patients has been found to be 1.5-7 times higher than in patients with adequate nutrition[10-14]. Malnourished hip fracture patients are also less likely to return to baseline functioning, undergo longer hospitalizations, and are more likely to suffer complications such as pressure ulcers, urinary tract infections (UTIs), delirium, pulmonary complications, and surgical site infections and sepsis[7,10-12,15-17]. Nutritional status can therefore serve as an interventional target to improve hip fracture outcomes.
Nutritional interventions have been evaluated in prior studies, but these studies are highly variable and limited[18]. None have been shown to effectively influence mortality. There is evidence suggesting that preoperative nutritional assessments, dietary counseling, and supplements (e.g. vitamin D, calcium) can lead to decreased complication rates and shorter hospital stays[19-22]. Oral nutritional supplementation (ONS) has also been shown to reduce hospital length and complications[18,23]. Despite the relative ease of administering ONS, older adult patients may not be able or willing to consume it[24]. Moreover, factors such as delirium-related confusion and/or uncooperative behavior and opioid-induced sedation and/or constipation in the post-injury/postoperative period can further act as barriers to administration of ONS. Nasogastric tube (NGT) feeding can address these limitations. Prior studies have attempted NGT feeding as an intervention for hip fracture patients[25-27], and although it did show promise, the studies were underpowered and difficult to execute.
Considering the potential of NGT feeding and the limitations of ONS in older adult hip fracture patients, it would be worthwhile to evaluate the effect of early NGT feeding on hip fracture outcomes in a randomized, controlled, prospective fashion. However, from prior literature and our experience, feasibility may be questionable. We therefore initiated a prospective longitudinal pilot study with the aim of evaluating feasibility of initiating early enteral nutrition via NGT in patients undergoing hip fracture surgery. We also sought to describe and compare outcomes between patients that underwent early NGT feeding and patients that received standard of care.
Methods
This was a prospective longitudinal pilot study conducted at a level 1 trauma center in a metropolitan area between June 2019 and January 2020. An Institutional Review Board (IRB) approved this study in 2019 (study ID: A18-419). Patients aged ≥65 that suffered an isolated acute hip fracture (i.e. femoral neck, intertrochanteric, subtrochanteric) due to low-energy trauma and underwent surgery within 48 hours of admission were eligible for inclusion. Exclusion criteria included high-energy trauma, acetabular and/or pelvic fractures, periprosthetic fractures, nonunion or malunion, pathologic fractures, active cancer diagnosis, immunosuppression therapy and/or chronic steroid use, over 12 hours on the ground after fall/injury, organ failure, history of malabsorptive disorder or milk protein allergy, and hospice enrollment.
Eligible patients were identified using ICD-10 codes followed by chart review and patient interviews. Patients were divided into two groups: (1) those receiving NGT intraoperatively and (2) those receiving standard of care (oral intake with calorie counts). Recruitment was targeted towards the interventional group with an option for patients to be in the control group if they refused intervention. A standardized consent form approved by the IRB was used for all patients. All patients received a standardized educational video approved by the study staff via tablet on the background and rationale of the study as well as on the risks and benefits for NGT placement and feeding. Education was preferentially administered in the presence of patient family.
Regarding the interventional group, in-house registered dieticians (RDs) monitored the interventional protocol, which involved early enteral nutrition starting from the time of surgery, when the tube was placed, up until either 3 weeks postop or until patients met appropriate nutritional intake by mouth. Regarding the control group, patients were followed throughout their hospitalization with a calorie count and were provided ONS to encourage intake per standard of care. Additional nutritional needs for all patients were assessed by the RDs as well as the primary physicians.
Variables of interest included demographics, Charlson Comorbidity Index (CCI)[28], injury characteristics, surgical characteristics, baseline mobility and functional data, and fat/muscle wasting severity among others. Ambulation was classified as “Independent”, “Assisted Cane”, “Assisted Walker”, or “Does not Walk”. Living arrangement status was classified as “Independent”, “Assisted Living”, or “SNF” (skilled nursing facility). Muscle/fat wasting was classified as “None”, “Mild”, “Moderate”, and “Severe”.
For our primary aim, case report forms (CRFs) were generated and used to record patient and staff comments regarding the intervention. Particularly for the intervention group, data regarding NGT-related events such as early removal and complications were also recorded. For our secondary aim, we planned to record outcomes of interest at discharge, 30 days, 90 days, and one year after for mortality, readmissions, complications, ambulation status, weight, muscle/fat wasting, and hand grip strength.
Study aims were purely descriptive, for which power analysis was unnecessary. Based on prior institutional observation and needs and limitations deemed reasonable by the study team, we aimed to enroll a total of 60 patients (30 intervention, 30 control) in the study. The study was discontinued early due to significant limitations and low feasibility.
Results
A total of 16 patients were recruited (7 intervention, 9 control). The cohort had an average age of 78.0 ± 11.9 and was mostly female (11/16, 68.8%). Average BMI and CCI were 24.3 ± 3.5 and 0.8 ± 1.2, respectively. There were 10 femoral neck fractures, 5 intertrochanteric fractures, and 1 subtrochanteric fracture. Surgery occurred 23.3 ± 11.0 hours after presentation and included total hip arthroplasty (5, 31.3%), hemiarthroplasty (3, 18.8%), intramedullary nailing (6, 37.5%), and percutaneous screw fixation (2, 12.5%). Patients were functional at baseline, with 14 patients (87.5%) living independently and 14 patients (87.5%) ambulating with or without assistive devices. Average baseline weight was 70.5 ± 17.4 kg. Eight patients (57.1%) were identified as at-risk for malnutrition, while 5 patients (31.3%) were already malnourished. Most patients had some degree of wasting (12, 80.0%).
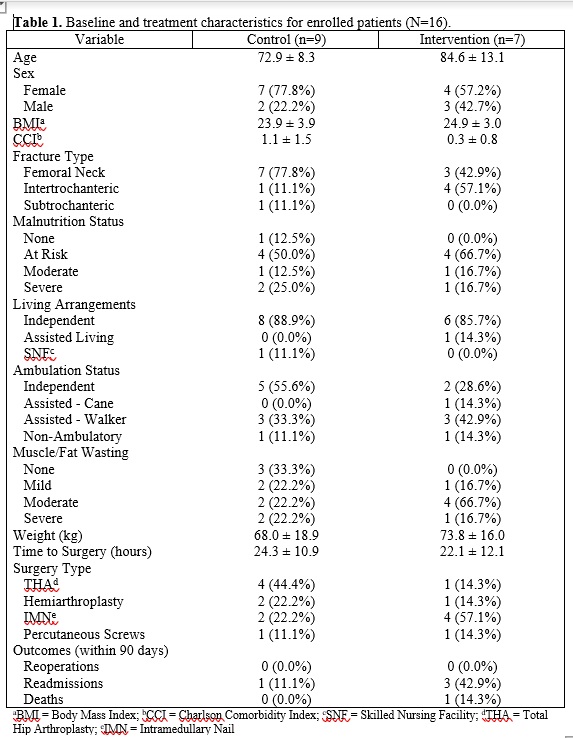
The majority of patients decided to forgo NGT and enter the control group. All patients expressed discomfort with the idea of having a NGT during their postoperative period, using descriptions including words such as “disturbing”, “squeamish”, and “ick” among others. One patient declined due to prior negative experience with a NGT. Regarding the 7 patients in the intervention arm, one patient never received a NGT intraoperatively due to provider-authorized order cancellation with unknown reason. All NGT patients underwent tube removal at an average of 3.3 days (0-11 days). Five of 6 were removed per request due to intolerable symptoms including general discomfort (most common), dysphagia, cough, irritation/pain, and nausea/vomiting. One patient removed the NGT inadvertently during an episode of delirium. There were no complications with tube maintenance, but one patient had a malpositioned NGT that remained misplaced after an attempted advancement. The patient requested tube removal and withdrew from the study, citing frustration with the experience as a reason for withdrawal.
There were no reoperations within 90 days after discharge. A total of 4 patients (25.0%) experienced readmissions within 60 days of discharge for reasons including gastrointestinal bleeding, hypotension, uncontrollable leg pain, and cardiopulmonary failure. One readmitted patient (6.3%) passed away within 10 days after discharge. We were unable to collect enough meaningful data for other outcome measures or evaluate patterns between the study arms.
Discussion
Malnutrition is associated with worse outcomes following hip fracture. Effective nutritional interventions have yet to be identified. NGT feeding has been evaluated in the past via studies subject to limitations and barriers to implementation. This pilot study thus assessed feasibility of implementing early NGT feeding. Although most patients were at-risk of malnutrition, the majority refused to partake in the intervention. Patients cited mostly discomfort and negative connotation associated with NGT as reasons to forgo intervention. Within the intervention group, all patients had their NGTs removed per request, inadvertently, or via clinical staff decision-making. We were unable to collect sufficient meaningful outcome data.
Prior Work Regarding NGT Feeding in Hip Fracture Patients
Prior studies have evaluated the effect of tube feeding in hip fracture patients. Bastow et al. identified 744 females ages 68-91, classified them into 3 groups based on arm bulk (“well-nourished”, “thin”, and “very thin”), and noted that “thinner” patients had lower voluntary food intake, longer rehabilitation time, and higher mortality. They then randomized 122 patients from the thin and very thin groups into either standard of care or overnight supplementary NGT feeding (1000 kcal, 28 g of protein) starting within 5 days postoperatively. They found that the intervention patients experienced improvements in albumin and pre-albumin as well as shorter hospitalization length and rehabilitation time. Within the very thin group, mortality for tube-fed patients was 8% compared to 22% for controls, but this did not reach significance due to low statistical power (N=48). The authors noted that 22% patients were unable to tolerate NGT[27].
Sullivan et al. conducted a non-blinded randomized controlled trial of just 18 patients to evaluate the safety and efficacy of aggressive postoperative enteral nutrition via NGT (125 cc/hour over 11 hours) versus standard of care. The authors determined the intervention was safe and reported higher nutrient intake in the intervention group, no group differences in complications, and reduction in six-month mortality for the NGT group (0% vs 50%). They noted that all patients were willing to participate in the intervention and were successfully fed over an average of 2 weeks[25]. A subsequent pilot study involving more centers and patients aged ≥64 was conducted in order to confirm these preliminary results. Although the authors recruited 57 patients (30 control, 27 intervention), 5 patients in the intervention group were never started on NGT feeding due to refusal or inability to receive NGT. Of the 22 patients started on NGT feeding, 20 underwent NGT removal either inadvertently or per request. Only 5 patients received relatively consistent NGT feedings until the study endpoint of six months[26].
In contrast to Bastow et al. and Sullivan et al., the majority of our patients refused NGT feeding. All intervention patients had their NGT removed, most frequently per patient request, within 10 days of placement. We were unable to collect meaningful data to describe outcomes and observe trends between the groups. Consistent with prior literature, NGT intolerance, among other reasons, was cited as a reason for discontinuation of the intervention.
Study Feasibility: Barriers and Potential Solutions
Considering our findings and the literature, we were able to identify key barriers that could serve as targets to improve feasibility of a prospective NGT feeding study for hip fractures (Table 2). One of the main barriers is stigma perceived by patients, family, and staff that is associated with tube feeding. Not only is tube feeding uncomfortable, but it is also conventionally seen as an indicator of end-of-life. Bastow et al. had commented in their study that most of the prospective tube feeding studies at the time were conducted on patients with metastatic cancer[27]. In our pilot study, most patients that refused intervention reported apprehension and hesitance to the idea of NGT placement and/or maintenance. Patients mainly commented on perceived physical and psychological discomfort with the tube. Although our study did not specifically aim to ask patients for their refusal reasoning, it is likely that this perception influenced willingness to participate in the study intervention. We attempted to mitigate for this by implementing comprehensive NGT feeding education in the presence of family and clinicians. However, it is possible that this stigma can also emanate from the patient family or the involved clinical staff (e.g. physicians, nurses, RDs), which may have influenced the patient’s decision to enroll in the intervention. We regardless still believe it is important to provide patient education to improve implementation success. That being said, it is possible that an institution-wide culture shift on tube feeding may be necessary to attain study feasibility.
The high rate of NGT discontinuation is a notable barrier. Patient-requested tube removals comprised most of our discontinuations. Consistent with the reports of Sullivan et al. and Bastow et al., these occurred mostly due to symptomatic intolerance. It is also possible that the aforementioned stigma may have influenced patients to choose NGT discontinuation. We also found that one of the intervention patients never received the NGT due to an unexplained order cancellation by a provider. Although we were unable to ascertain the reason, it is likely that the provider was unaware of the study. Considering these observations, a strategy to mitigate tube discontinuation could involve therapeutic symptomatic support to help patients cope with the discomfort of having a NGT. Enhanced intra- and inter-departmental communication can also help raise study awareness and prevent unnecessary tube removals by staff. Additionally, tube bridling could be a useful strategy to ensure higher tube compliance.
Tube compliance and study feasibility may be enhanced by selectively targeting hip fracture patients more willing and able to undergo NGT placement and feeding. The hip fracture patient population is heterogeneous and has been found to exhibit different outcomes based on baseline patient characteristics such as age and comorbidities[29]. While Sullivan et al. randomized all recruited patients, Bastow et al. targeted patients for intervention based on anthropometric measurements. This strategy may have enhanced feasibility and follow-up for this study compared to Sullivan et al., evidenced by the 78% of patients[27] versus 19% of patients[26] that continued tube feeding until the study endpoint. It should be noted that targeting a specific subset of the population may introduce selection bias, thus diminishing study quality. However, for feasibility and initial investigations, this strategy may be helpful.
Sample is a commonplace barrier in the literature that was observed in our study. Bastow et al. did not detect a statistical difference between intervention and control in the very thin group despite the observable effect (8% vs 22%)[27]. Similar to Sullivan et al., we were unable to reach target sample . Furthermore, Sullivan et al. commented that, to replicate the effect seen in their prior study[25], approximately 1700 patients would have to be enrolled[26]. As was observed by Sullivan et al. and in our study, patient refusal and unplanned NGT removal can significantly decimate sample . Therefore, implementing strategies to tackle the aforementioned barriers of stigma and tube removals can address the barrier of sample . Despite the difficulty of conducting them, large multicenter trials may also help circumvent this barrier.
Institutional and staffing logistics can limit study feasibility. Implementation and standardization of study protocols and tube feeding/maintenance methodology would be highly beneficial, as it has shown to lead to enhanced compliance and outcomes pertaining to patients receiving tube feeding[30-32]. Designation of an administrative body dedicated to ensuring protocol implementation/standardization could potentially help not just at the institutional level, but among centers as well. This could help facilitate multicenter studies that can increase study sample .
In addition to protocol implementation and standardization, staff education on feeding tube rationale, placement, and maintenance is crucial, as prior studies have reported on a significant prevalence of tube malpositioning and tube-related complications as well as a low prevalence of quality nutrition administration via enteral nutrition therapy[33-35]. For one patient in our pilot study, the NGT was malpositioned and required multiple readjustments, which initially failed. This influenced the patient enough to choose to discontinue the NGT and withdraw from the study prior to another readjustment attempt. From our experience, we believe that enhancing staff training in tube placement and maintenance, via education or other means, would be a good target to improve tube compliance, diminish tube removals, and increase study feasibility.
One beneficial takeaway from our pilot study worth mentioning is the importance of multidisciplinary care and interdepartmental collaboration. This study included participation from the orthopaedic surgery, geriatric medicine, hospital medicine, and nutrition teams. From our experience, patients seemed to appreciate the sense of being cared for by a large team comprised of multiple areas of expertise. This is consistent with prior work that has noted that older adult hip fracture patients tend to perceive “confidence that they are receiving best care” and “access to the provider” as top priorities compared to other aspects such as physical outcomes[36]. Regarding study feasibility, it is possible that enhanced interdepartmental collaboration could have a positive effect on patients that may lead to increased tube compliance or tolerance. Additionally, it could increase the institutional awareness of the study and ensure better standardization of study protocols.
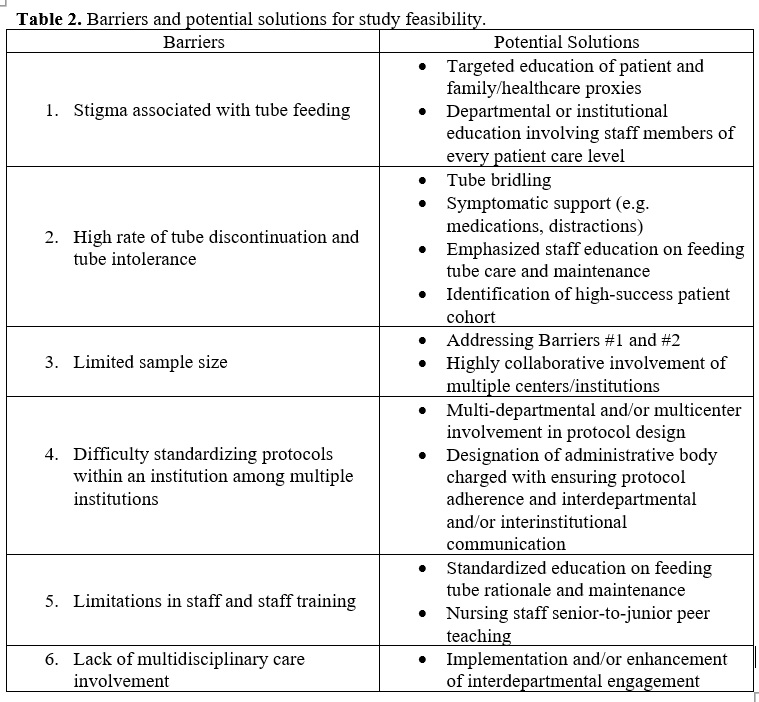
In summary, our prospective longitudinal study was halted due to implementation barriers. The most notable barriers found in our study and in the literature include large required sample and frequent interventional discontinuation. The perceived stigma of tube feeding seems to also play a role in study feasibility at least in our institution. We were able to propose some solutions to the aforementioned barriers, with the enhancement of multidisciplinary care and interdepartmental collaboration as a possible overarching benefit.
Conclusion
A randomized prospective trial evaluating early aggressive enteral tube feeding versus standard of care can be challenging to conduct. Our pilot study failed due to barriers specific to patients, clinical staff, and study environment that prevented the investigators from collecting long-term meaningful data. Potential solutions were identified including enhancement of interdepartmental collaboration.
Disclosure Statement
Julie A. Switzer MD is a committee member for the American Academy of Orthopaedic Surgeons (AAOS), the American Orthopaedic Associations (AOA), and the International Geriatric Fracture Society (IGFS). She is also on the editorial board of Geriatric Orthopaedic Surgery & Rehabilitation. All other authors reported no disclosures or conflicts of interest.
Data Availability Statement
This work does not contain any private or non-accessible research materials. Data can be made available upon reasonable request.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
- Arshi A, Rezzadeh K, Stavrakis AI, et al. Standardized hospital-based care programs improve geriatric hip fracture outcomes: an analysis of the ACS NSQIP targeted hip fracture series. J Orthop Trauma. 2019;33(6):e223-e228.
- Cooper C, Cole Z, Holroyd C, et al. Secular trends in the incidence of hip and other osteoporotic fractures. Osteoporos Int. 2011;22(5):1277-1288.
- Huang Z, Himes JH, McGovem PG. Nutrition and subsequent hip fracture risk among a national cohort of white women. Am J Epidemiol. 1996;144(2):124-134.
- Schousboe JT, Paudel ML, Taylor BC, et al. Pre-fracture individual characteristics associated with high total health care costs after hip fracture. Osteoporos Int. 2017;28(3):889-899.
- Burge R, Dawson‐Hughes B, Solomon DH, et al. Incidence and economic burden of osteoporosis‐related fractures in the United States, 2005–2025. J Bone Miner Res. 2007;22(3):465-475.
- Kates SL, Shields E, Behrend C, et al. Financial implications of hospital readmission after hip fracture. Geriatr Orthop Surg Rehabil. 2015;6(3):140-146.
- Malafarina V, Reginster J-Y, Cabrerizo S, et al. Nutritional status and nutritional treatment are related to outcomes and mortality in older adults with hip fracture. Nutrients. 2018;10(5):555.
- Koren-Hakim T, Weiss A, Hershkovitz A, et al. The relationship between nutritional status of hip fracture operated elderly patients and their functioning, comorbidity and outcome. Clin Nutr. 2012;31(6):917-921.
- Nematy M, Hickson M, Brynes A, et al. Vulnerable patients with a fractured neck of femur: nutritional status and support in hospital. J Hum Nutr Diet. 2006;19(3):209-218.
- Pimlott BJ, Jones CA, Beaupre LA, et al. Prognostic impact of pre-operative albumin on short-term mortality and complications in patients with hip fracture. Arch Gerontol Geriatr. 2011;53(1):90-94.
- Bohl DD, Shen MR, Hannon CP, et al. Serum albumin predicts survival and postoperative course following surgery for geriatric hip fracture. J Bone Joint Surg Am. 2017;99(24):2110-2118.
- Koval KJ, Maurer SG, Su ET, et al. The effects of nutritional status on outcome after hip fracture. J Orthop Trauma. 1999;13(3):164-169.
- Schaller F, Sidelnikov E, Theiler R, et al. Mild to moderate cognitive impairment is a major risk factor for mortality and nursing home admission in the first year after hip fracture. Bone. 2012;51(3):347-352.
- Miyanishi K, Jingushi S, Torisu T. Mortality after hip fracture in Japan: the role of nutritional status. J Orthop Surg. 2010;18(3):265-270.
- Baumgarten M, Margolis DJ, Orwig DL, et al. Pressure ulcers in elderly patients with hip fracture across the continuum of care. J Am Geriatr Soc. 2009;57(5):863-870.
- Fry DE, Pine M, Jones BL, et al. Patient characteristics and the occurrence of never events. Arch Surg. 2010;145(2):148-151.
- Banks M, Bauer J, Graves N, et al. Malnutrition and pressure ulcer risk in adults in Australian health care facilities. Nutrition. 2010;26(9):896-901.
- Avenell A, Smith TO, Curtain JP, et al. Nutritional supplementation for hip fracture aftercare in older people. Cochrane Database Syst Rev. 2016 (11).
- Lai W-Y, Chiu Y-C, Lu K-C, et al. Beneficial effects of preoperative oral nutrition supplements on postoperative outcomes in geriatric hip fracture patients: A PRISMA-compliant systematic review and meta-analysis of randomized controlled studies. Med. 2021;100(47).
- Williams DG, Ohnuma T, Haines KL, et al. Association between early postoperative nutritional supplement utilisation and length of stay in malnourished hip fracture patients. Br J Anaesth. 2021;126(3):730-737.
- Ekinci O, Yanık S, Terzioğlu Bebitoğlu B, et al. Effect of calcium β‐hydroxy‐β‐methylbutyrate (CaHMB), vitamin D, and protein supplementation on postoperative immobilization in malnourished older adult patients with hip fracture: a randomized controlled study. Nutr Clin Pract. 2016;31(6):829-835.
- Malafarina V, Uriz-Otano F, Malafarina C, et al. Effectiveness of nutritional supplementation on sarcopenia and recovery in hip fracture patients. A multi-centre randomized trial. Maturitas. 2017;101:42-50.
- Anbar R, Beloosesky Y, Cohen J, et al. Tight calorie control in geriatric patients following hip fracture decreases complications: a randomized, controlled study. Clin Nutr. 2014;33(1):23-28.
- Milne AJ, Woolford H, Mason J, et al. Early diagnosis of dementia by GPs: an exploratory study of attitudes. Aging Ment Health. 2000;4(4):292-300.
- Sullivan DH, Nelson CL, Bopp MM, et al. Nightly enteral nutrition support of elderly hip fracture patients: a phase I trial. J Am Coll Nutr. 1998;17(2):155-161.
- Sullivan DH, Nelson CL, Klimberg VS, et al. Nightly enteral nutrition support of elderly hip fracture patients: a pilot study. J Am Coll Nutr. 2004;23(6):683-691.
- Bastow M, Rawlings J, Allison S. Benefits of supplementary tube feeding after fractured neck of femur: a randomised controlled trial. Br Med J (Clin Res Ed). 1983;287(6405):1589-1592.
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373-383.
- Penrod JD, Litke A, Hawkes WG, et al. Heterogeneity in hip fracture patients: age, functional status, and comorbidity. J Am Geriatr Soc. 2007;55(3):407-413.
- Furlong-Dillard JM, Miller BJ, Sward KA, et al. The association between feeding protocol compliance and weight gain following high-risk neonatal cardiac surgery. Cardiol Young. 2019;29(5):594-601.
- Na S, Lee H, Koh SO, et al. Physician compliance with tube feeding protocol improves nutritional and clinical outcomes in acute lung injury patients. Korean J Crit Care Med. 2010;25(3):136-143.
- Holt B, Graves C, Faraklas I, et al. Compliance with nutrition support guidelines in acutely burned patients. Burns. 2012;38(5):645-649.
- Nogueira DA, Ferreira LP, de Lúcia RPA, et al. High Frequency of Non-Compliance with Quality Indicators of Enteral and Parenteral Nutritional Therapy in Hospitalized Patients. Nutrients. 2020;12(8):2408.
- Ellett MLC. What is known about methods of correctly placing gastric tubes in adults and children. Gastroenterol Nurs. 2004;27(6):253-259.
- Sorokin R, Gottlieb JE. Enhancing patient safety during feeding‐tube insertion: a review of more than 2000 insertions. J Parenter Enteral Nutr. 2006;30(5):440-445.
- Turner N, Dinh JM, Durham J, et al. Development of a Questionnaire to Assess Patient Priorities in Hip Fracture Care. Geriatr Orthop Surg Rehabil. 2020;11:2151459320946009.
Required Disclosures and Declaration
Copyright Information: No Copyright Information Added
IRB Approval Information: Yes
Disclosure Information:


